Research
Mechanisms of mechanosensing and cellular decision-making
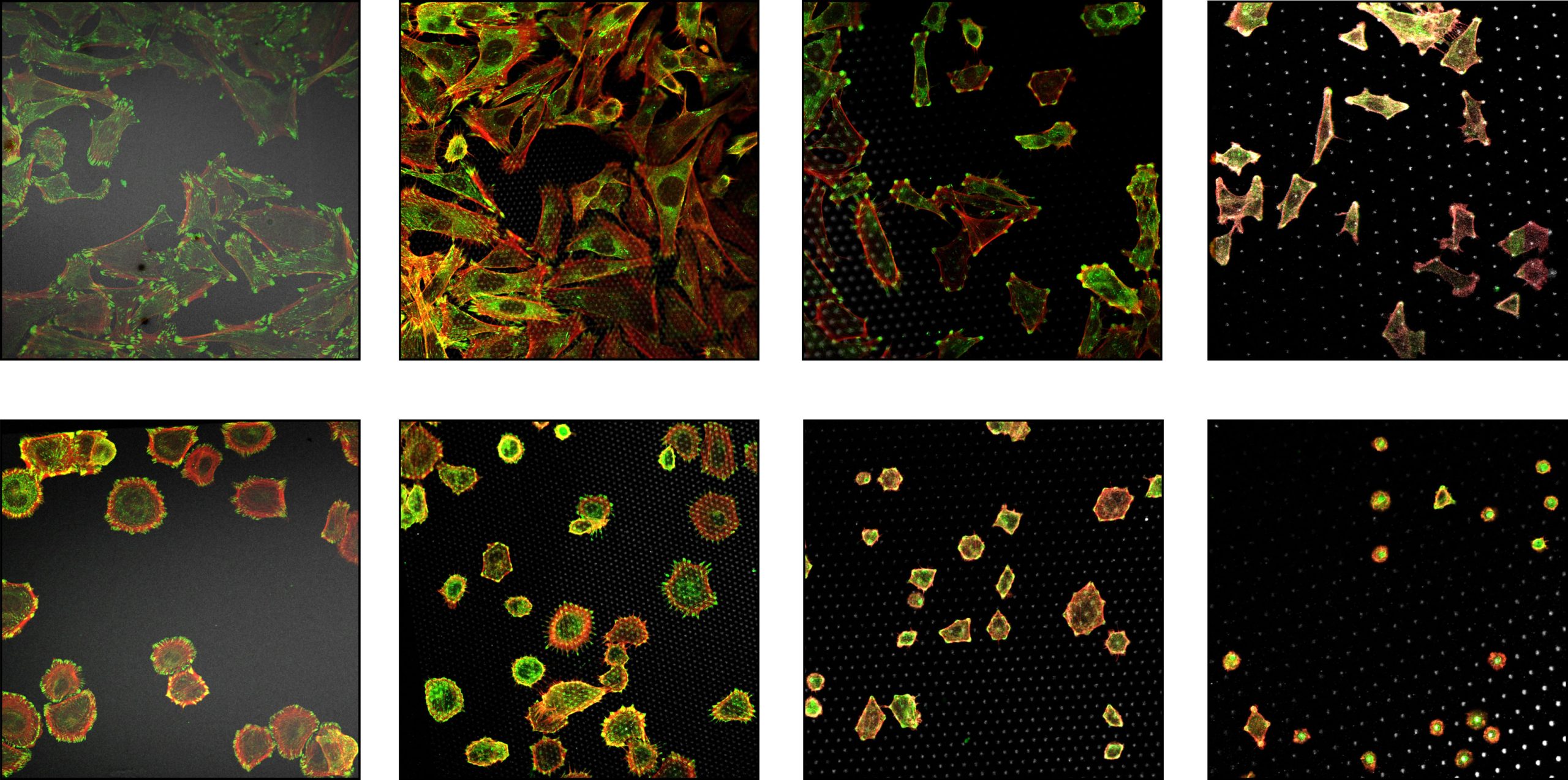
Since the very early days of cell biology studies, more than 100 years ago, researchers recognized the importance of cell attachment to rigid surfaces and its essential role in growth and migration. Later it was recognized that unlike normal tissue cells, cancer cells are able to grow in suspension without solid support, making them “anchorage‐independent”. This hallmark property of cancer cells has highlighted the importance of correct sensing of the mechanical properties of the extracellular matrix (ECM). In particular, ECM rigidity has emerged as a major determinant of many cellular aspects.
Our recent work (Feld et al., 2020) uncovered that cells probe the rigidity of their surrounding matrix through actomyosin-driven displacements that are surprisingly independent of the matrix’s mechanical properties. This finding reveals that the forces generated during these probing events are inherently non-mechanosensitive. Importantly, these events are not passive mechanical reactions; they act as critical checkpoints in cellular decision-making, shaping fundamental behaviors such as growth, migration, apoptosis, and differentiation. Building on these insights, we aim to understand how cells integrate mechanical cues across space and time to orchestrate these complex fate decisions. By elucidating these mechanisms, we hope to uncover new principles of mechanobiology that could inform innovative therapeutic strategies for diseases where these processes are disrupted, including cancer, fibrosis, and aging.