Research
Coordination Between Cell–Matrix and Cell–Cell Adhesions in Cell Migration
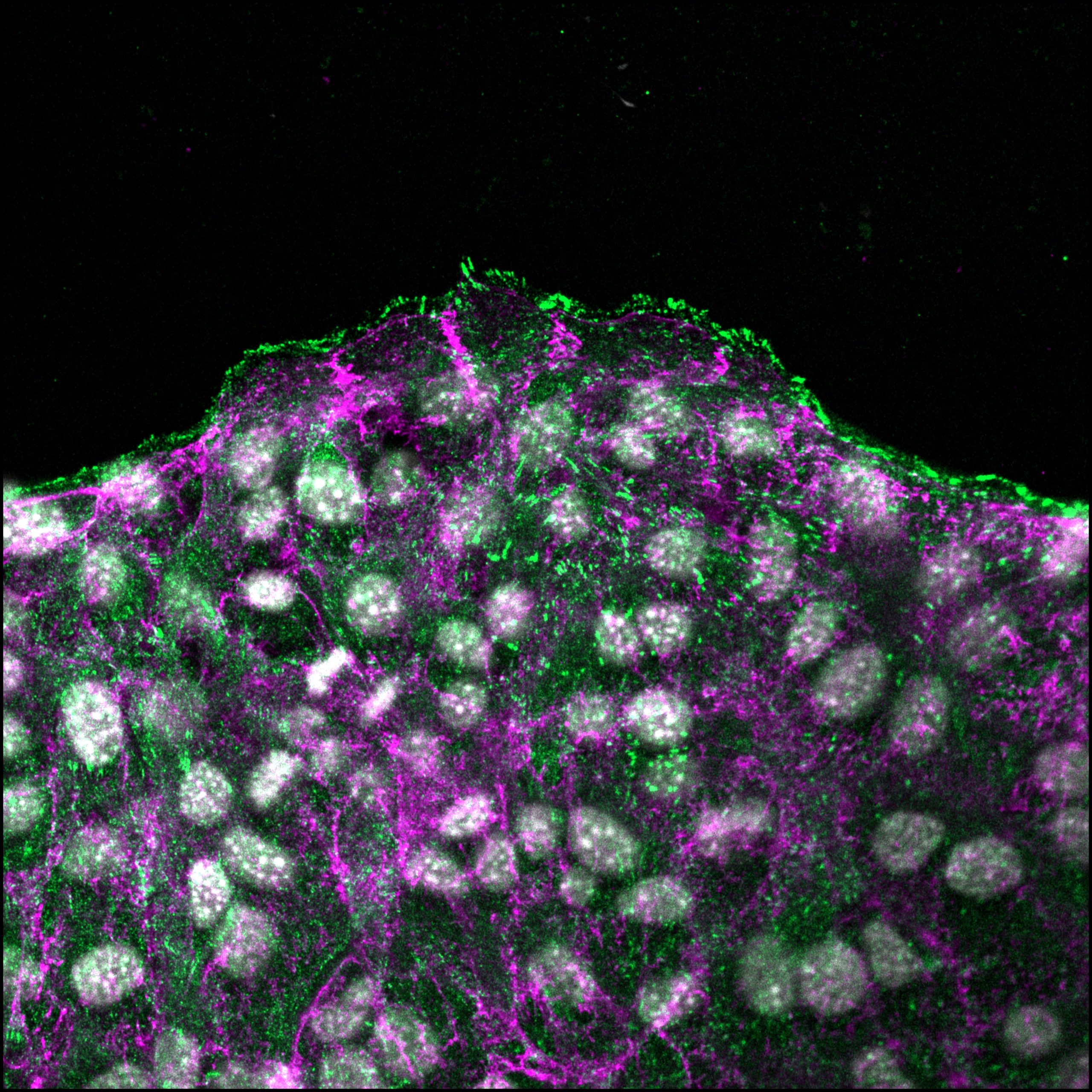
Our recent findings reveal that collective cell migration is a highly efficient mechanism for cancer metastasis, challenging the long-standing paradigm that single-cell invasion following epithelial-to-mesenchymal transition (EMT) is the dominant route of metastatic spread. Unlike solitary cells that rely primarily on cell–matrix interactions to navigate through the extracellular environment, collectively migrating cells maintain cell–cell junctions while simultaneously engaging with the matrix, enabling them to move as cohesive units. This dual adhesion strategy provides mechanical stability, directional persistence, and enhanced survival during invasion.
We are particularly interested in dissecting how cell–cell and cell–matrix adhesions coordinate during collective migration, as both adhesion systems are indispensable for this process. Intriguingly, several key proteins are shared between these adhesion complexes, suggesting a dynamic redistribution of molecular components as cells transition between different migratory modes. Our data indicate a spatiotemporal interplay between these structures in pancreatic ductal adenocarcinoma, where collective invasion appears to confer advantages in overcoming stromal barriers and resisting apoptosis.
By integrating advanced live-cell imaging, traction force microscopy, and molecular perturbation approaches, we aim to uncover how mechanical forces and signaling pathways are balanced between cell–cell and cell–matrix adhesions during collective migration. Understanding these mechanisms will not only redefine current models of metastatic dissemination but also identify novel therapeutic targets to disrupt collective invasion – a strategy that may prove more effective than interventions aimed solely at EMT-driven single-cell migration.