Research
Cell–Matrix Interactions in Fibrosis
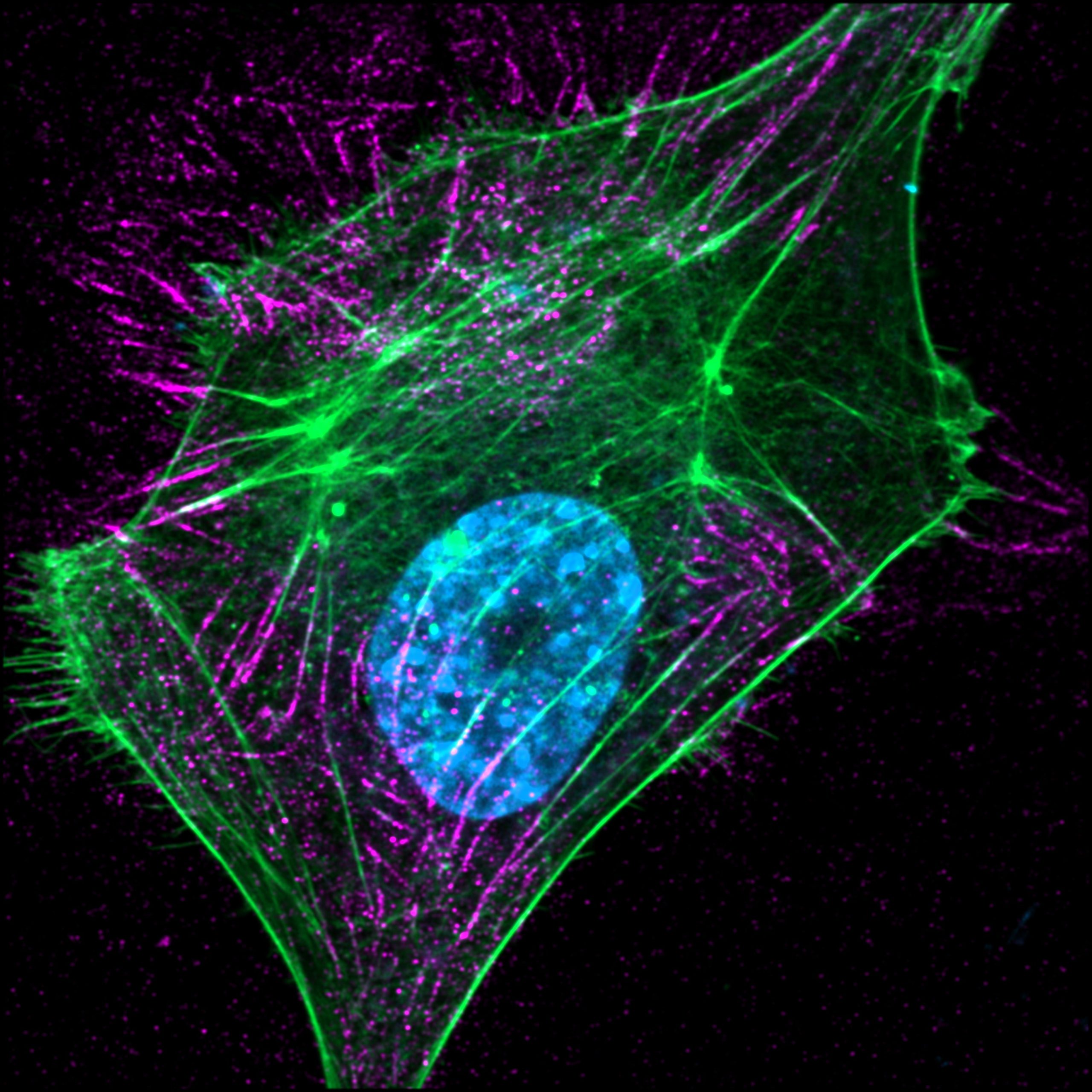
Fibrosis is characterized by excessive deposition and remodeling of the extracellular matrix (ECM), leading to tissue stiffening and impaired organ function. We recently uncovered a novel regulatory mechanism that controls ECM assembly through enzymatic oxidation of fibronectin, in collaboration with Prof. Peleg Hasson’s lab (Melamed et al. 2023). This finding provides critical insight into how fibronectin fibrillogenesis is orchestrated during fibrotic progression and highlights a previously unrecognized layer of molecular regulation in tissue remodeling.
Building on this discovery, we have developed a new class of fibronectin fibrillogenesis inhibitors designed to disrupt pathological ECM assembly. These inhibitors are currently being evaluated for their anti-fibrotic and anti-scarring potential in both in vitro and in vivo models. By targeting the early steps of fibronectin network formation, this approach offers a promising strategy to modulate ECM dynamics and restore tissue homeostasis.
Our long-term vision is to translate these findings into clinically relevant therapies for fibrotic diseases, which remain a major unmet medical need across multiple organs, including the lungs, liver, and heart. If successful, these inhibitors could pave the way for innovative interventions that not only halt fibrosis but also improve regenerative outcomes following injury or surgery.